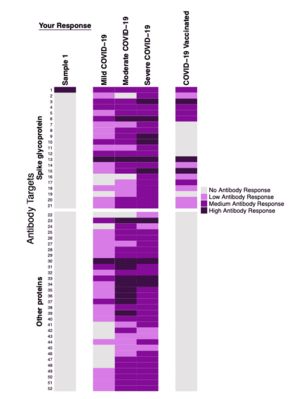
Serimmune antibody analysis results
| Project Infobox | |
|---|---|
| Self researcher(s) | User:Madprime, User:Sprague |
| Related tools | |
| Related topics | |
Builds on project(s) |
|
| Has inspired | Projects (0) |
The Serimmune COVID-19 Study is a longitudinal research study conducted by the Serimmune company. The study uses the company's "Serum Epitope Repertoire Analysis (SERA) technology" to study antibody responses, especially in the context of COVID-19 vaccination and exposure.
The study returns research-grade results (not intended for diagnostic clinical use) to participants. As described in the study consent document, these results are not clinically validated and not meant to serve as a clinical assay. They are, nevertheless, interesting to look at!
The study is planned to last five years, with blood samples collected every six months. Samples are self-collected by participants using a home collection kit and sent by mail.
How Serimmune's Technology WorksEdit
According to the company's pre-published[1] research, the technology uses a combination of biochemical assays and a machine learning-based analytics pipeline.
First, they chemically break a blood sample into one million molecules they call the "epitope repertoire". Roughly speaking, this is an approximation of possible ways that antibodies can bond in this individual.
Next, they compare this individual's epitope with others taken from people with known exposure to specific infections. Theoretically, this is a map to those molecules associated with a particular pathogen. Using a machine learning algorithm trained on epitopes from known infections, the company generates a report to indicate the similarity with this person's epitope.
The gold standard for antibody measurement is a complex process called ELISA (enzyme-linked immunosorbent assay) that involves smudging a bit of sample onto a specially-prepared plate full of proteins known to be associated with the disease. The plate is designed to change color if an antibody to the disease happens to match whatever’s on the plate.
Serimmune claims their test works very well compared to ELISA. It has a high “specificity” (true negative): when they say a sample doesn’t have COVID, they’re almost always right. In fact, when they applied their test to samples taken before the pandemic, every single time they correctly ruled out COVID.
SARS-CoV-2 Sample ReportsEdit
| Individual | Date | Report | Vaccination | Natural infection | Notes |
|---|---|---|---|---|---|
| User:Sprague | 2021-12-14 | File:RichardSpragueSerimmuneCOVID 20211214.pdf | Pfizer | No | Pfizer vaccine (two shots, no booster) |
| User:Madprime | 2022-02-03 | File:TOEAIX 20220203.pdf | Moderna | No | Moderna vaccine (two shots, plus booster). |
| User:Madprime | 2022-05-27 | File:TOEAIX 20220527.pdf | Moderna | Yes | Covid positive on 2022-05-01. |
| User:Madprime | 2022-08-04 | File:TOEAIX 20220804.pdf | Moderna | Yes | No additional history. |
| User:Igerg | 2022-04-13 | File:EXACZL 20220413.pdf | Moderna | No | Moderna vaccine (two shots, plus booster) - chronic spontaneous urticaria following booster |
| User:EricJain | 2022-07-21 | File:EricJain-Serimmune-20220721.pdf | Pfizer | No | Report includes comparison to 2022-01-20. Vaccinations: 2021-04-21 (Pfizer), 2021-05-12 (Pfizer), 2021-12-08 (Pfizer). |
Other resultsEdit
In addition to COVID-19 related antibody analyses, the study shares research analysis of antibodies related to infectious diseases (Lyme Disease, Ehrlichiosis, Anaplasmosis, Babesiosis, Chagas Disease) and autoimmune conditions (Celiac Disease).
| Individual | Date | Report | Notes |
|---|---|---|---|
| User:Sprague | 2021-12-14 | File:RichardSpragueSerimmune 20211214.pdf | |
| User:Madprime | 2022-02-03 | File:TOEAIX Expanded 20220203.pdf | No medical history related to these analyses. |
| User:Igerg | 2022-04-13 | File:EXACZL Additional 20220413.pdf | Tested for Lyme several times, all negative |
| User:Madprime | 2022-05-27 | File:TOEAIX Additional 20220527.pdf | |
| User:Madprime | 2022-08-04 | File:TOEAIX Additional 20220804.pdf |
- ↑ High-resolution mapping and characterization of epitopes in COVID-19 patients Winston A. Haynes, Kathy Kamath, Joel Bozekowski, Elisabeth Baum-Jones, Melissa Campbell, Arnau Casanovas-Massana, Patrick S. Daugherty, Charles S. Dela Cruz, Abhilash Dhal, Shelli F. Farhadian, Lynn Fitzgibbons, John Fournier, Michael Jhatro, Gregory Jordan, Debra Kessler, Jon Klein, Carolina Lucas, Larry L. Luchsinger, Brian Martinez, Mary C. Muenker, Lauren Pischel, Jack Reifert, Jaymie R. Sawyer, Rebecca Waitz, Elsio A. Wunder Jr., Minlu Zhang, Yale IMPACT Team, Akiko Iwasaki, Albert I. Ko, John C. Shon medRxiv 2020.11.23.20235002; doi: https://doi.org/10.1101/2020.11.23.20235002